 We showed that an OsTIR1(F74G) mutant demonstrated no detectable basal degradation and that depletion could be induced at about a 670 times lower concentration of a ligand, 5-phenyl-indole-3-acetic acid (5-Ph-IAA). Here, we show AID version 2 (AID2) system that overcomes all these drawbacks of the original AID system. Multiple groups, including our own, recently reported a method to suppress basal degradation 21, 22, 23, but a high dose of IAA was still used in all cases. As far as we know, the AID system has not been successfully applied to adult mice. Indeed, IAA is known to cause kidney toxicity when it is converted to indoxyl sulphate in the liver 20. This could be a potential problem for applying the original AID system to stem cell lines and mice. These have no acute short-term effect in yeast and cancer cell lines, but we noted that some cell lines showed slow growth when cultured for long-term with 500 µM IAA (data will be shown later). Second, the required doses of indole-3-acetic acid (IAA), a natural auxin used for degradation, are relatively high (typically 100–500 µM). In such cases, we needed to control the expression of OsTIR1 using a conditional tetracycline-inducible promoter. In some cases, we were not successful in fusing mAID to proteins essential for viability in cells that constitutively expressed OsTIR1. First, leaky degradation of mAID-fused proteins (hereafter referred to as basal degradation) was observed even without auxin 15. This AID technology has recently been used in many functional studies because of its high efficiency and rapid depletion 16, 17, 18, 19. We and others showed that, in many cases, mAID-fused proteins can be degraded in human cells expressing OsTIR1 with a half-life (T1/2) of 20–40 min 12, 14, 15. We identified a 7-kD degron derived from Arabidopsis IAA17, that we termed mini-AID (mAID) 13. When expressed in non-plant cells, Oryza sativa TIR1 (OsTIR1) forms a Skp1–Cul1–F-box (SCF) E3 ligase complex with endogenous components. 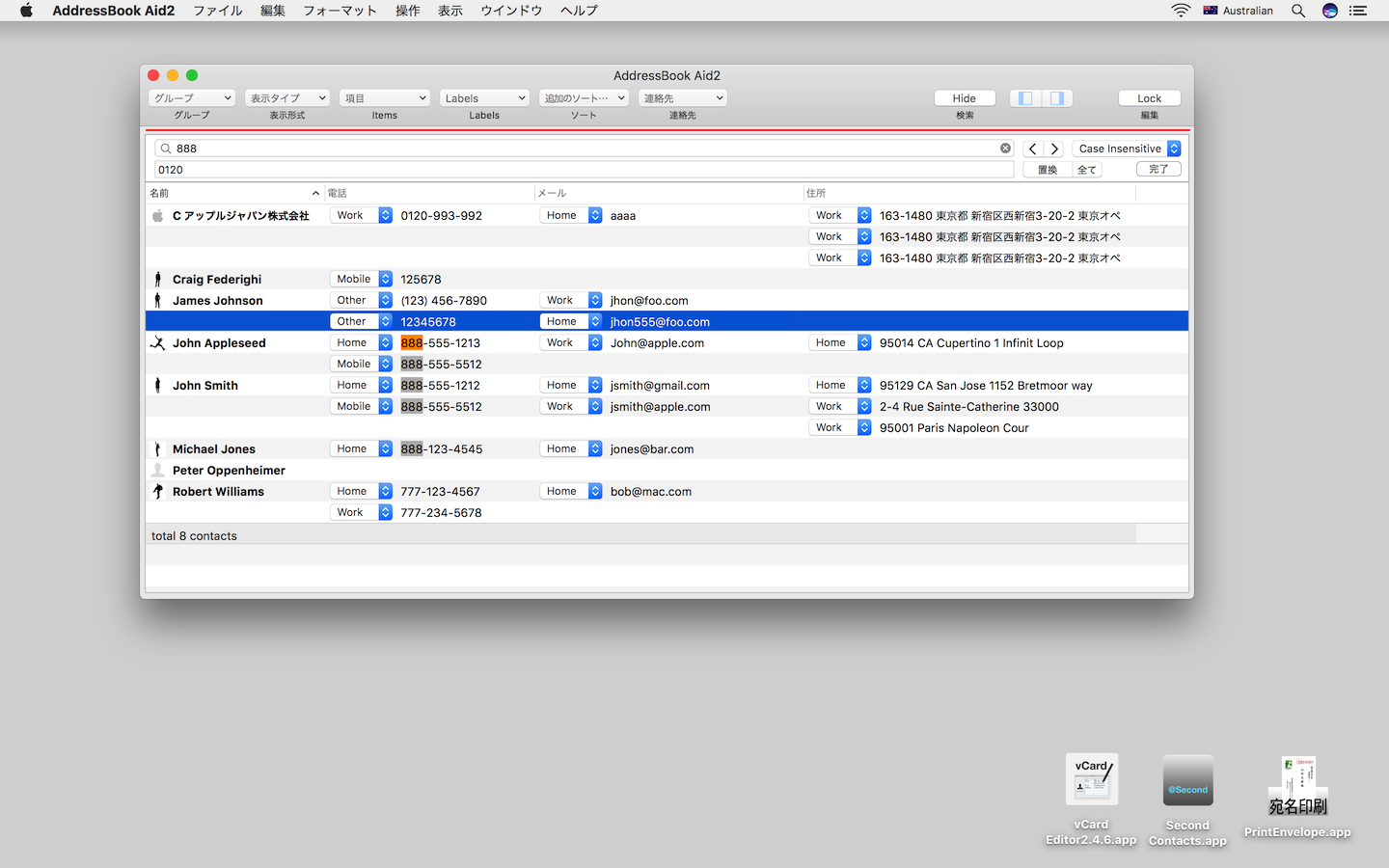 
We pioneered development of one of the major degron-based systems, auxin-inducible degron (AID) technology 12. A more general approach has been developed, which uses a small tag (called degron) that induces degradation in the presence or absence of a defined ligand, so that the level of degradation of a degron-fused protein via the ubiquitin–proteasome pathway can be rapidly controlled by ligand administration 6, 7, 8, 9, 10, 11. However, to employ these methodologies, a specific PROTAC or antibody must be developed for each protein of interest. For this purpose, heterobifunctional chemical degraders 4 (known as proteolysis-targeting chimeras or PROTACs) or antibodies (e.g., Trim-Away 5) can be used. Protein knockdown can be achieved by recruiting a protein of interest to an E3 ubiquitin ligase. Proteins can be targeted for rapid degradation by the ubiquitin–proteasome pathway 3. To achieve rapid depletion, protein-knockdown systems are becoming more popular. However, these technologies are not ideal for studying highly dynamic processes, such as cell cycle, differentiation or neural activity, because of the slow rate of depletion of the protein of interest. Conditional gene knockout 1 or siRNA-based mRNA depletion 2 has been employed in many studies. An ideal conditional depletion should be achieved rapidly and efficiently before the resultant phenotype is complicated or compromised by secondary effects and/or adaptation. Studies of protein function in living cells and animals are greatly assisted by the conditional depletion of a protein of interest. We demonstrate successful generation of human cell mutants for genes that were previously difficult to deal with, and show that AID2 achieves rapid target depletion not only in yeast and mammalian cells, but also in mice. AID2, which employs an OsTIR1(F74G) mutant and a ligand, 5-Ph-IAA, shows no detectable leaky degradation, requires a 670-times lower ligand concentration, and achieves even quicker degradation than the conventional AID. Here, we overcome these problems by taking advantage of a bump-and-hole approach to establish the AID version 2 (AID2) system. These negative features make it difficult to control precisely the expression level of a protein of interest in living cells and to apply this method to mice. However, the current AID system has two major drawbacks: leaky degradation and the requirement for a high dose of auxin. 
Protein knockdown using the auxin-inducible degron (AID) technology is useful to study protein function in living cells because it induces rapid depletion, which makes it possible to observe an immediate phenotype. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
